How CO₂ Causes Corrosion in Carbon Steel Pipelines
CO₂ corrosion, often referred to as sweet corrosion, is one of the most common internal corrosion mechanisms affecting carbon steel pipelines in the oil and gas industry. Despite being widely studied and relatively well understood, CO₂ corrosion remains a frequent cause of unexpected pipeline degradation and failures.
The root cause is not a lack of knowledge, but a simplified understanding of how CO₂ actually leads to corrosion under real operating conditions. This article explains the fundamental mechanism of CO₂ corrosion in carbon steel pipelines and why its severity is often underestimated in integrity management.
CO₂ itself is not corrosive
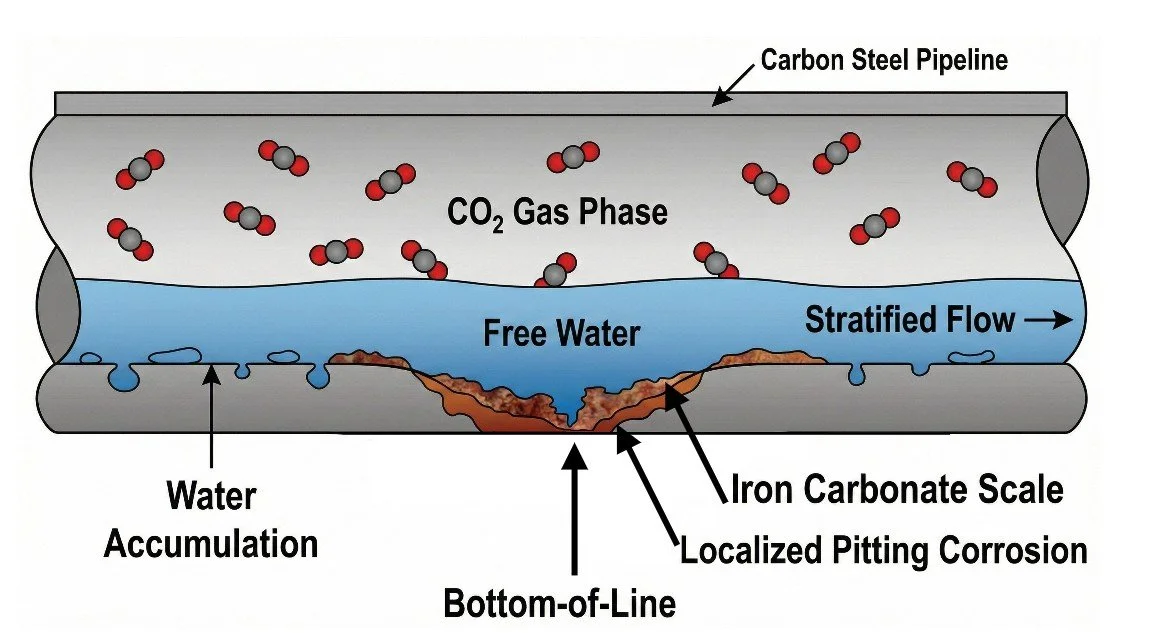
A common misconception is that CO₂ directly corrodes steel. In reality, dry CO₂ is not corrosive to carbon steel.
Corrosion only occurs when three conditions are met:
CO₂ is present,
free water is present at the pipe wall,
electrochemical reactions are allowed to proceed.
This principle is consistently reflected in corrosion mechanism descriptions such as those in ISO 15156, which explicitly treats CO₂ corrosion as an aqueous-phase phenomenon, not a gas-phase one.
Formation of carbonic acid in free water
When CO₂ dissolves in free water, it forms carbonic acid (H₂CO₃):
CO₂ + H₂O ⇌ H₂CO₃
Although carbonic acid is a weak acid, it significantly lowers pH and accelerates the anodic dissolution of iron. The resulting electrochemical reactions lead to:
iron dissolution at the anode,
hydrogen evolution at the cathode,
progressive metal loss at the pipe wall.
The presence, chemistry, and residence time of free water therefore govern CO₂ corrosion severity far more than CO₂ concentration alone.
Why CO₂ corrosion is often localized
In real pipelines, free water rarely wets the pipe wall uniformly. Instead, it:
accumulates at low points,
flows intermittently in stratified or slug flow,
settles at the 6 o’clock position.
This leads to localized CO₂ corrosion, even when average corrosion rates appear low. Such localization explains why CO₂ corrosion frequently results in pitting or preferential bottom-of-line attack rather than uniform thinning.
This behavior is explicitly acknowledged in DNV-RP-F101, which treats CO₂ corrosion as a localized threat requiring careful interpretation of inspection data.
The role of iron carbonate (FeCO₃) scales
As corrosion progresses, iron ions released from the steel may react with carbonate ions to form iron carbonate (FeCO₃) scales.
Depending on conditions, FeCO₃ can:
partially protect the steel surface,
reduce corrosion rates,
or break down and expose fresh metal.
The formation and protectiveness of FeCO₃ depend strongly on:
temperature,
CO₂ partial pressure,
flow shear stress,
water chemistry.
FeCO₃
This conditional behavior explains why CO₂ corrosion can appear stable for long periods and then suddenly accelerate when conditions change.
Why operating conditions dominate CO₂ corrosion
CO₂ corrosion severity is not fixed. It evolves with:
increasing water cut,
changes in flow velocity,
temperature variations,
operational transients.
Standards such as ISO 13623 explicitly require that internal corrosion mechanisms, including CO₂ corrosion, be reassessed as operating conditions evolve, rather than assumed constant over the pipeline life.
This dynamic nature is a key reason why early-life corrosion assessments often become non-conservative.
CO₂ corrosion versus corrosion allowance
CO₂ corrosion is often managed implicitly through corrosion allowance. This approach assumes:
relatively uniform corrosion,
stable corrosion rates,
predictable consumption of wall thickness.
In practice, localized CO₂ corrosion frequently violates these assumptions, making corrosion allowance an unreliable primary safeguard without active corrosion control and monitoring.
Integrity implications of CO₂ corrosion
Correct understanding of CO₂ corrosion directly impacts:
corrosion monitoring strategy,
pigging and water management philosophy,
inhibitor selection and performance assessment,
inspection coverage and defect assessment,
fitness-for-service decisions.
Treating CO₂ corrosion as a “known and controlled” mechanism without continuous reassessment is one of the most common integrity blind spots.
Conclusion
CO₂ causes corrosion in carbon steel pipelines only when free water is present, but when it does, the resulting degradation can be severe, localized, and highly sensitive to operating conditions.
Pipelines rarely fail because CO₂ corrosion is unknown.
They fail because its mechanism is oversimplified and its evolution underestimated.
A realistic understanding of how CO₂ corrosion actually develops is essential for sustainable pipeline integrity management.