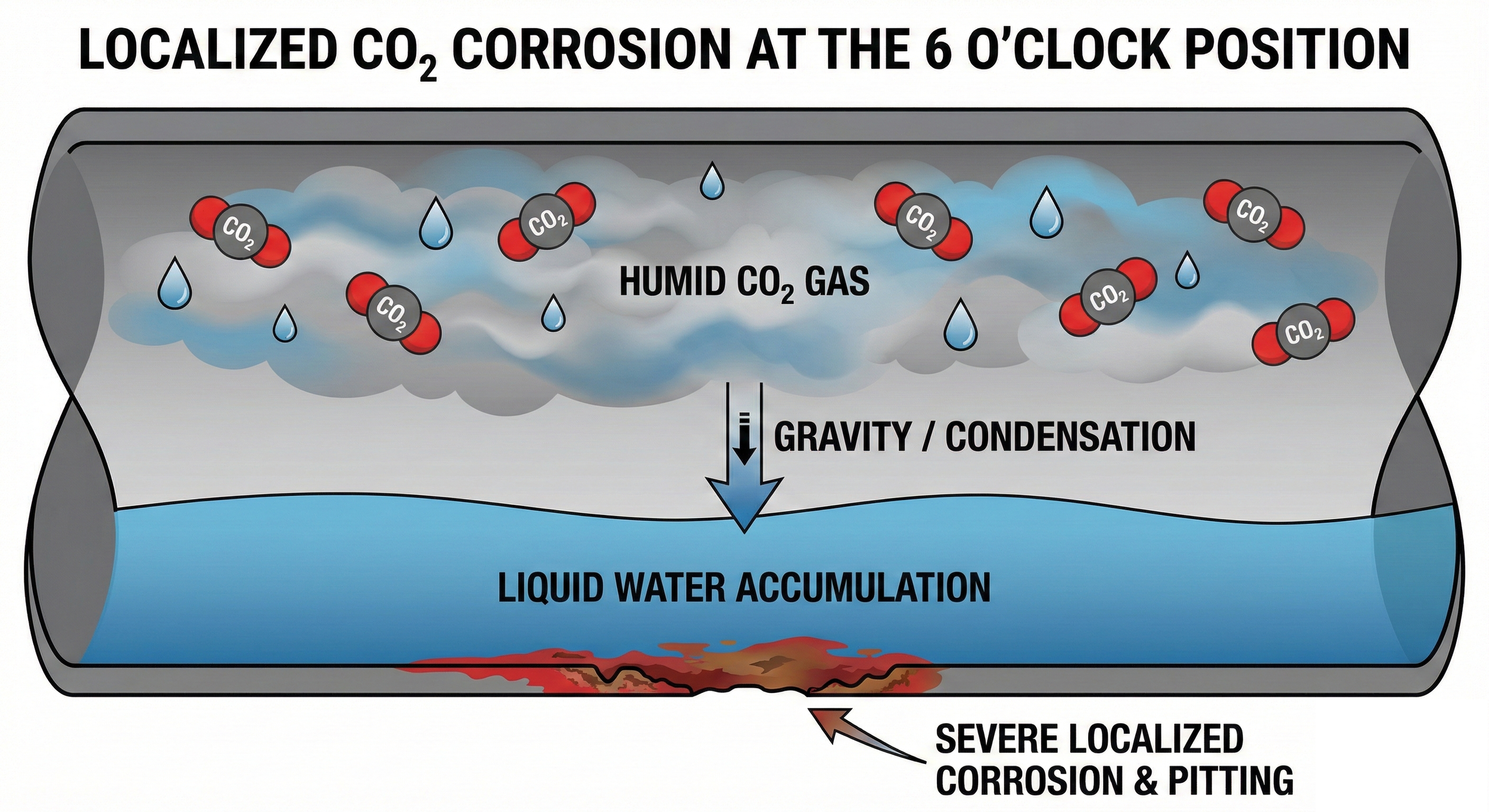
Why CO₂ Corrosion Often Concentrates at the 6 o’clock Position in Pipelines
One of the most consistent field observations in CO₂-corroded pipelines is the concentration of damage at the 6 o’clock position, i.e. the bottom of the pipe. This phenomenon is so common that it is sometimes treated as an empirical rule rather than a mechanistic outcome.
In reality, bottom-of-line CO₂ corrosion is not coincidental. It is the direct result of fluid behavior, flow regime, and corrosion chemistry interacting under real operating conditions. This article explains why CO₂ corrosion preferentially develops at the 6 o’clock position and why this localization is often underestimated in integrity assessments.
Gravity governs water behavior, not corrosion models
CO₂ corrosion requires free water. In multiphase pipelines, gravity inevitably causes free water to:
separate from the gas or oil phase,
migrate downward,
accumulate at the bottom of the pipe.
Even when the overall water cut is low, the bottom of the pipe experiences the longest and most continuous wetting time. Corrosion models assuming uniform wetting therefore fail to capture the true corrosion exposure at the 6 o’clock position.
This water-driven localization mechanism is implicitly recognized in corrosion descriptions used in API RP 571, which links CO₂ corrosion severity to aqueous phase behavior rather than bulk fluid composition.
Stratified and slug flow amplify bottom-of-line corrosion
In many oil and gas pipelines, flow is not fully turbulent and homogeneous. Common regimes include:
stratified flow,
intermittent (slug) flow,
low-velocity multiphase flow.
Under these conditions:
the upper pipe wall may remain intermittently dry,
the bottom remains continuously wetted,
CO₂ dissolves preferentially into the bottom water layer.
This creates a persistent electrochemical environment favorable to CO₂ corrosion at the 6 o’clock position, even when average corrosion indicators appear acceptable.
Local chemistry is more aggressive at the bottom
The aqueous phase at the bottom of the pipe often differs chemically from the bulk fluid:
higher CO₂ concentration due to longer residence time,
lower pH,
accumulation of iron ions and corrosion products,
possible presence of solids and deposits.
These conditions promote:
localized CO₂ corrosion,
under-deposit corrosion,
initiation of pitting beneath corrosion scales.
Such localization behavior is explicitly addressed in DNV-RP-F101, which treats bottom-of-line corrosion as a high-risk localized degradation mechanism.
FeCO₃ scales are least stable at the 6 o’clock position
While iron carbonate (FeCO₃) may form in CO₂ systems, its protectiveness is weakest at the bottom of the pipe due to:
continuous wetting,
solids accumulation,
flow-induced shear during slug passage,
scale detachment and redeposition.
Detached FeCO₃ often contributes to deposits that further shield the steel surface, creating conditions for accelerated under-deposit corrosion rather than protection.
This explains why FeCO₃-based assumptions frequently break down at the 6 o’clock position.
Why inspection and monitoring often miss bottom corrosion
Bottom-of-line corrosion is frequently underestimated because:
corrosion monitoring probes are not located at the lowest point,
spot UT measurements miss narrow pits,
average corrosion rates smooth out localized extremes,
ILI sizing uncertainty increases for narrow, deep defects.
Integrity programs that rely on average corrosion indicators often fail to detect critical 6 o’clock damage until advanced stages.
Operating conditions strongly influence 6 o’clock corrosion severity
The severity of bottom-of-line CO₂ corrosion is highly sensitive to:
flow velocity and shear stress,
pigging effectiveness,
water management and drainage,
operational transients (start-ups, shutdowns).
Standards such as ISO 13623 explicitly require operators to consider internal corrosion localization and its dependency on operating conditions when defining integrity strategies.
Integrity implications of 6 o’clock CO₂ corrosion
Concentration of corrosion at the 6 o’clock position has direct consequences for:
inspection strategy (coverage and technology),
corrosion growth rate determination,
fitness-for-service assessments,
remaining life calculations.
Uniform corrosion assumptions in the presence of bottom-of-line attack are structurally non-conservative.
How integrity engineers should address bottom-of-line corrosion
Effective mitigation and assessment include:
realistic flow regime analysis,
targeted inspection of low points,
optimized pigging and debris management,
reassessment of inhibitor distribution effectiveness,
conservative treatment of localized corrosion in FFS evaluations.
Bottom-of-line corrosion must be treated as a primary integrity threat, not a secondary effect.
Conclusion
CO₂ corrosion concentrates at the 6 o’clock position because that is where water, chemistry, and time combine most aggressively.
Pipelines rarely fail because CO₂ corrosion was unknown. They fail because localized bottom-of-line corrosion was averaged out, underestimated, or inspected too late.
Recognizing and managing 6 o’clock corrosion is therefore essential for credible pipeline integrity management.